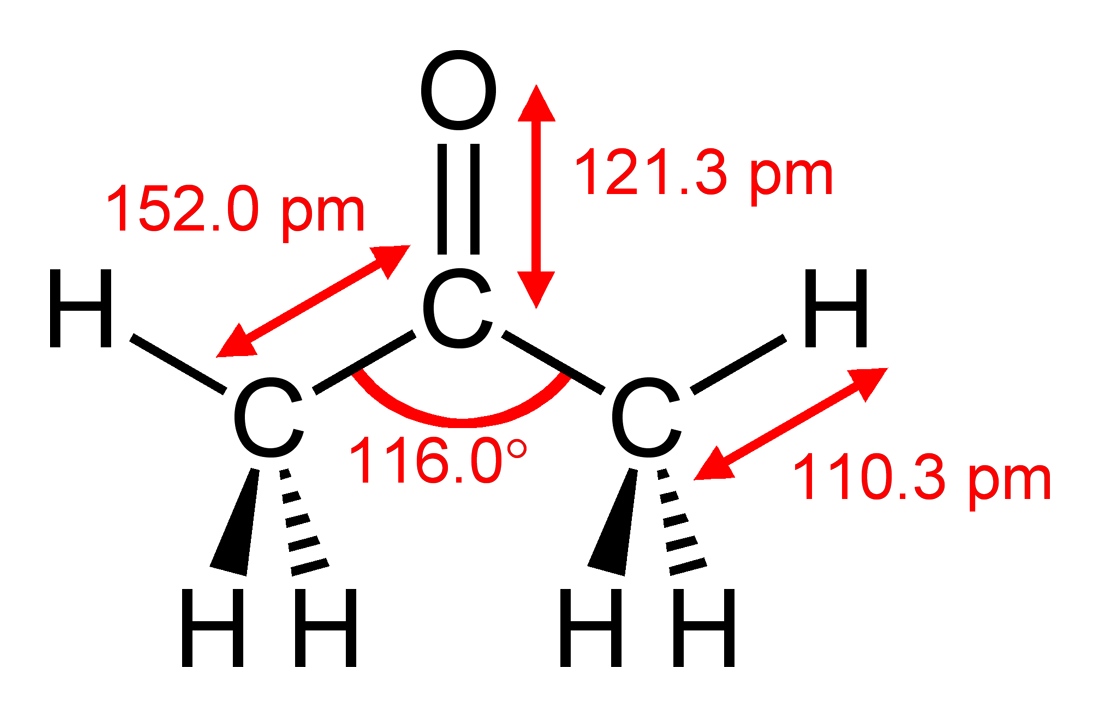
SOLVED: liquid density acetone 0.79 cm tetrahydrofuran 0.89 cm dimethyl sulfoxide 1.1 cm ethanolamine 1.0 cm methyl acetate 0.93 cm Next, the chemist measures the volume of the unknown liquid as 1.053

SOLVED: Consider the following thermochemical equation for the combustion of acetone, C3H6O, the main ingredient in nail polish remover: C3H6O(l)+4O2(g)→3CO2(g)+3H2O(g) ΔH∘rxn=−1790kJ If a bottle of nail polish remover contains 173 mL of