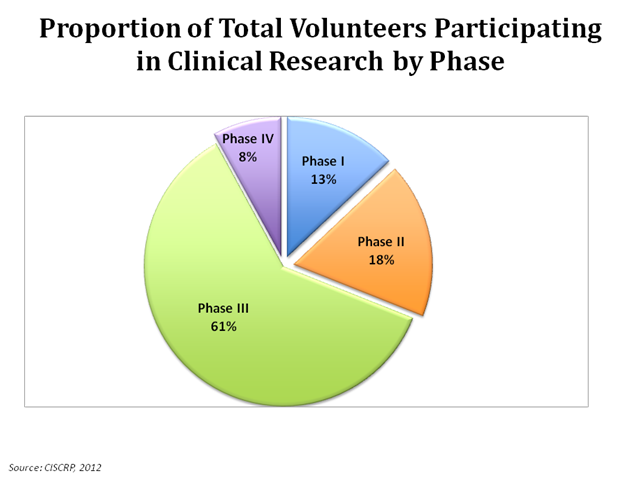
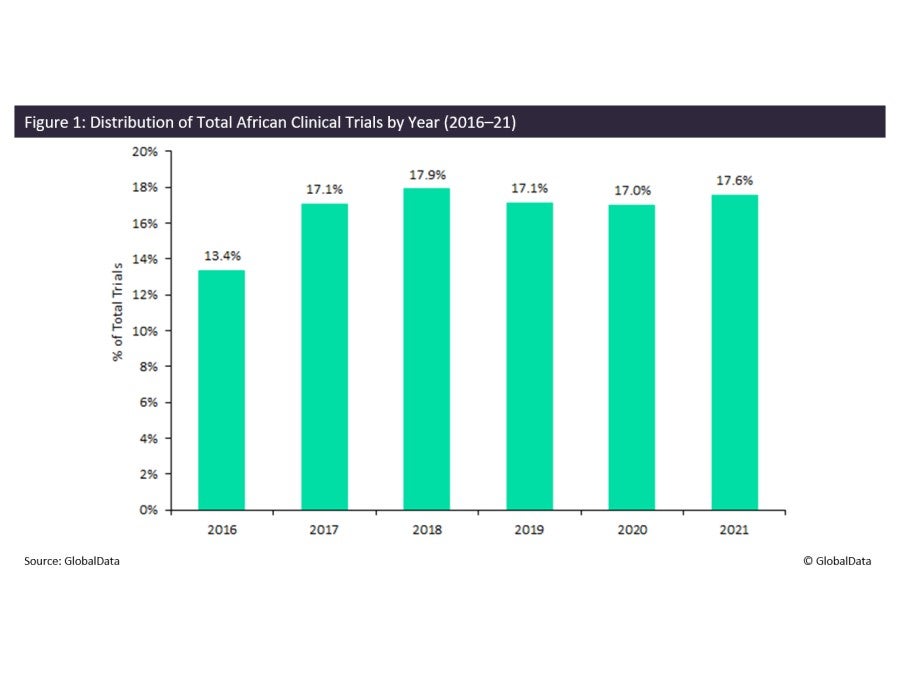
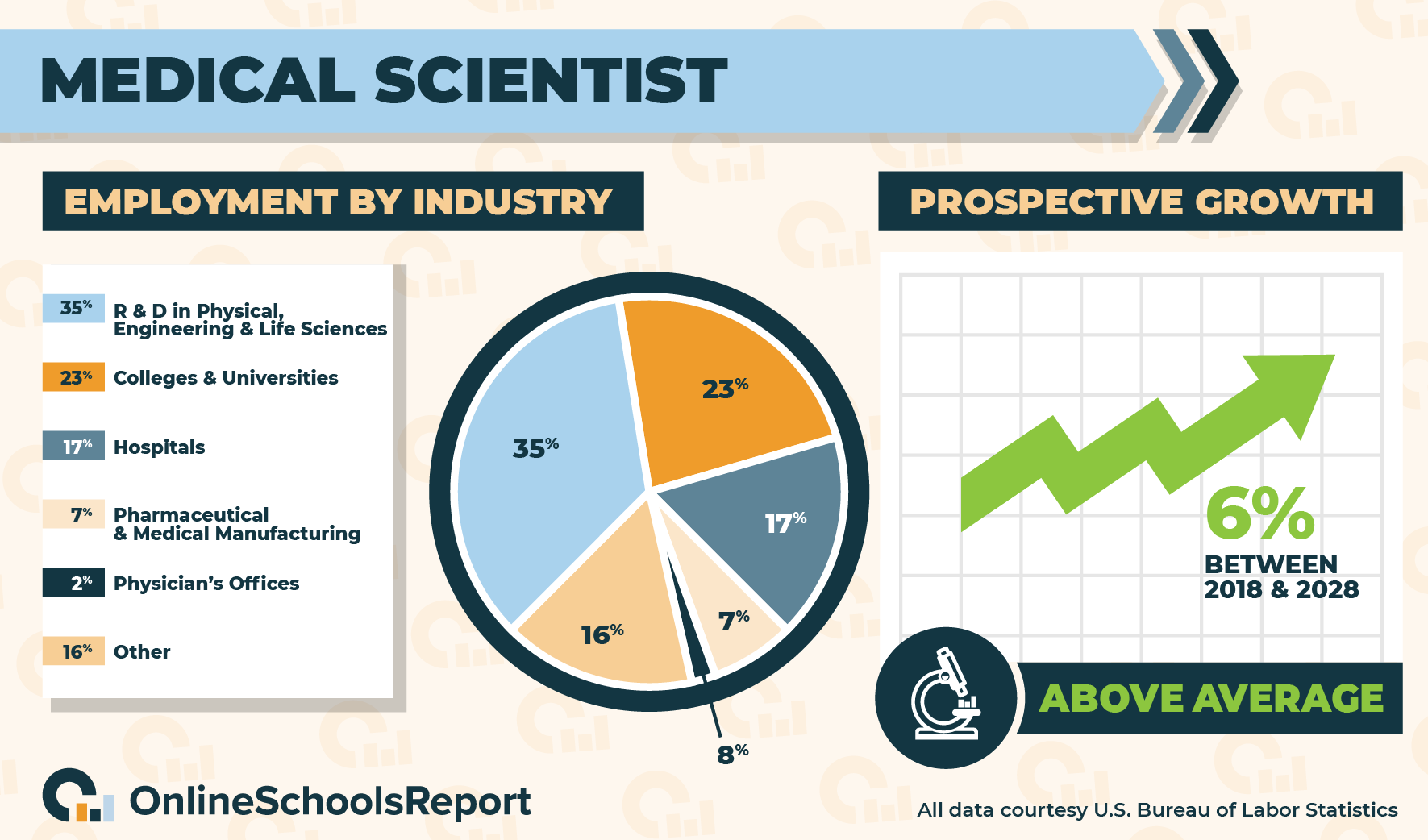
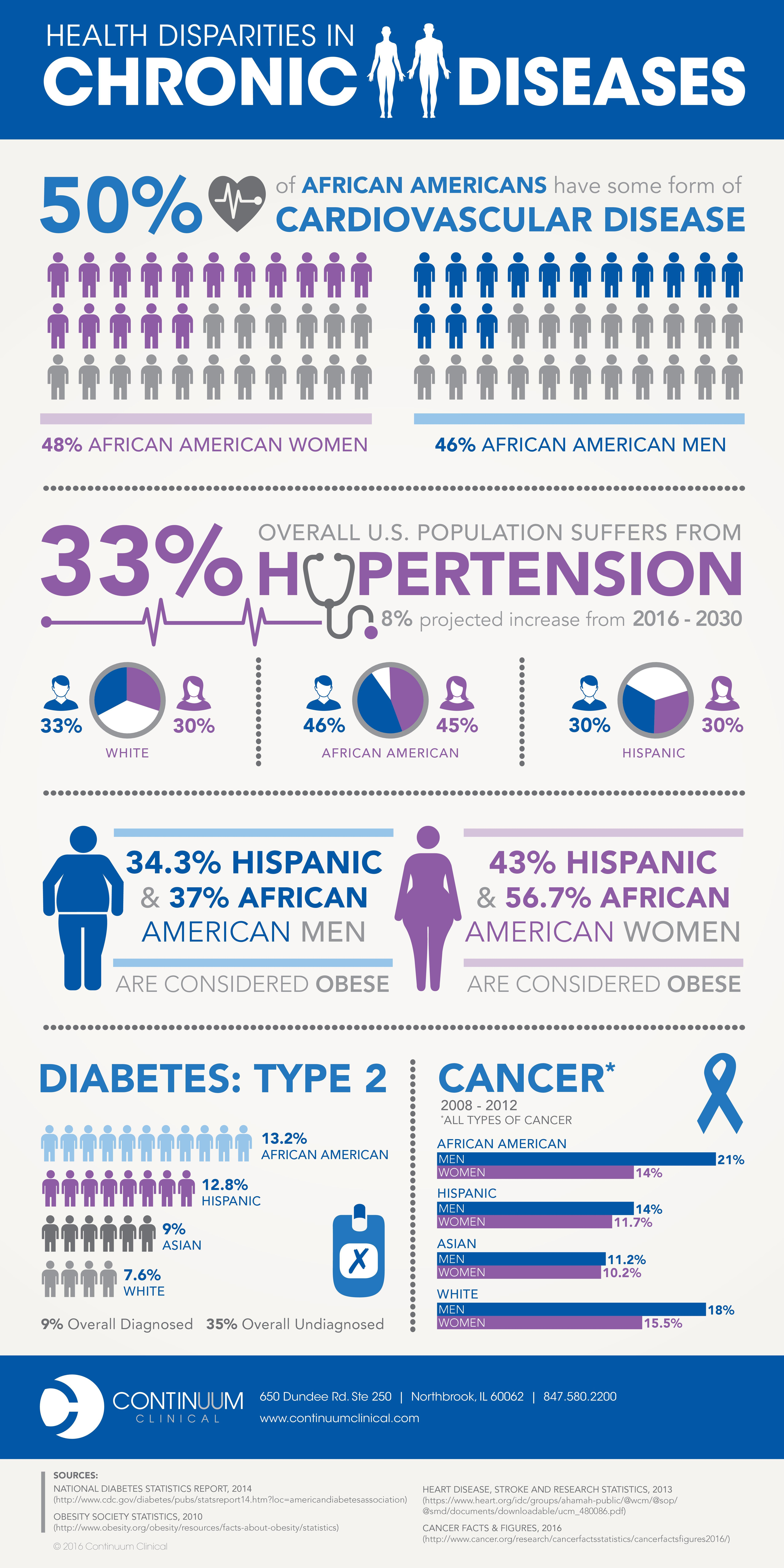
Enhancing pediatric clinical trial feasibility through the use of Bayesian statistics. | Pediatric clinical Trials, clinical research, clinical trials, medical trials, clinical cancer research, clinical studies, cro companies, phase 1 clinical trial,

Handbook for Clinical Research: Design, Statistics, and Implementation: 9781936287543: Medicine & Health Science Books @ Amazon.com

Increasing Diversity in Trials Requires Understanding Special Populations | 2019-12-15 | CenterWatch