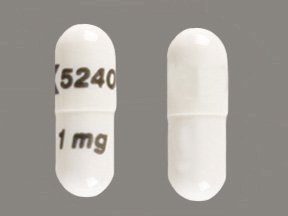
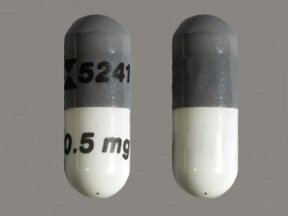
FDA announces recall of platelet-reducing medication due to risk of clotting or other adverse cardiovascular outcomes

These highlights do not include all the information needed to use ANAGRELIDE CAPSULES safely and effectively. See full prescribing information for ANAGRELIDE CAPSULES. ANAGRELIDE capsules, for oral useInitial U.S. Approval: 1997
Foglio illustrativo: Informazioni per il paziente Anagrelide Teva 0,5 mg capsule rigide anagrelide Medicinale equivalente Legga